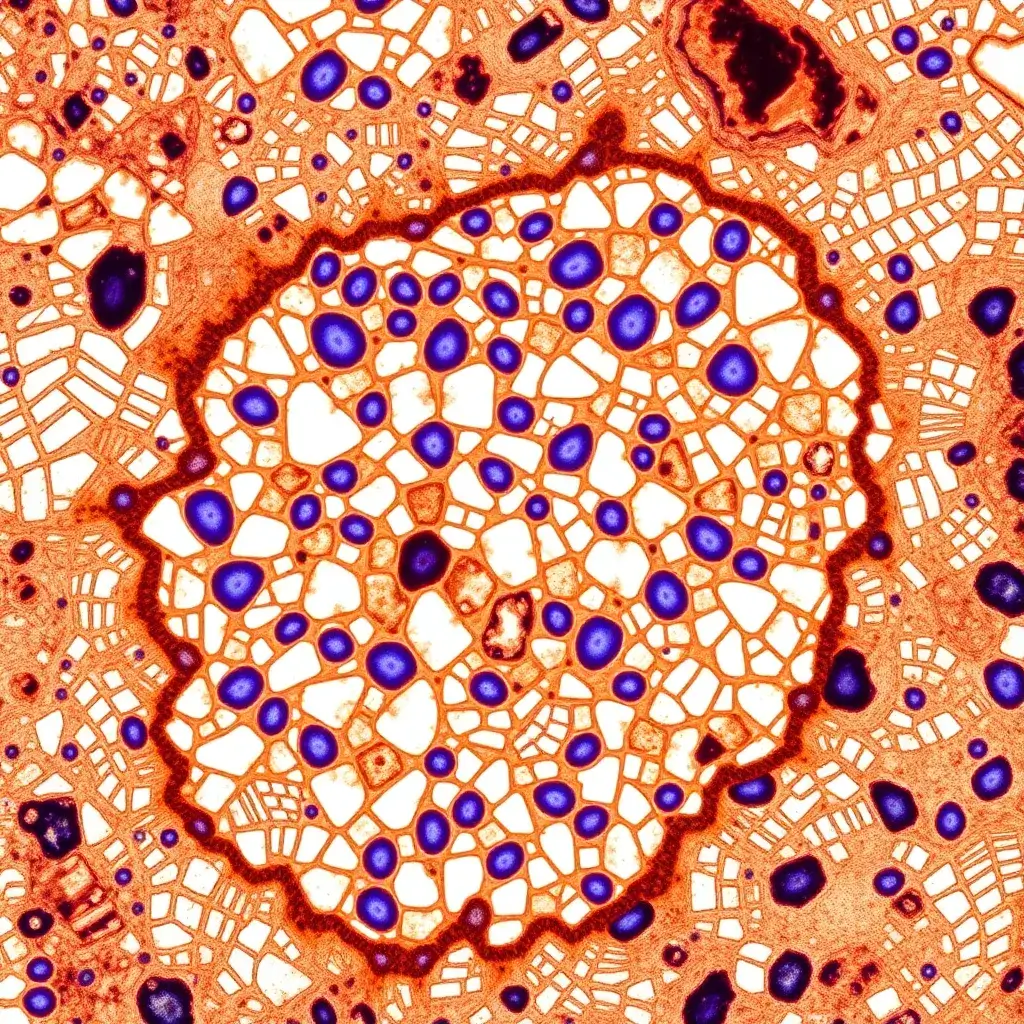
Immunohistochemistry
Immunohistochemistry (IHC) is a biochemical technique that utilizes antibodies to bind specifically to antigens within tissue sections, allowing for the detection of these antigens. Similarly, immunocytochemistry (ICC) is employed to identify antigens within individual cell layers. IHC is widely used in both research and clinical settings to visualize proteins, carbohydrates, and lipids of interest within tissues, including those found in cancerous tumors. IHC helps to identify specific molecular markers that indicate key cellular processes, such as proliferation or apoptosis (cell death). This technique is also extensively used in basic research to explore the distribution and localization of biomarkers, as well as differentially expressed proteins within various regions of biological tissue,
Overview
IHC Tissue Preparation, Antigen Retrieval, and Pretreatment
For immunohistochemistry (IHC) analysis, tissue samples are typically fixed to preserve cellular structures, embedded in paraffin for easy sectioning, and then sliced into thin sections using a microtome. After the initial tissue preparation, a crucial step is antigen retrieval. This step is required to break the protein cross-links formed during the fixation process, which can obscure antigenic sites on the proteins, making them inaccessible to antibodies. Antigen retrieval helps to expose these hidden epitopes, allowing for effective antibody binding.
The conditions for antigen retrieval and pretreatment must be determined empirically because the accessibility of the antigenic epitope may vary based on biological factors, such as the tissue type or antigen involved. Some antigens may require more aggressive treatment to “unmask” their binding sites, while others may not require any pretreatment at all. Common antigen retrieval methods include Heat-Induced Epitope Retrieval (HIER) and Proteolytic-Induced Epitope Retrieval (PIER). In HIER, heat is applied along with specialized buffers and enzymes, such as proteinase K, to facilitate the unmasking of antigens. On the other hand, PIER involves the use of various proteases, including proteinase K, trypsin, chymotrypsin, or pepsin, to digest the protein cross-links and expose the antigens.
IHC Staining and Detection
Once the tissue preparation and antigen retrieval steps are complete, the visualization of antibody-antigen interactions is the next critical phase in IHC. Several methods can be employed for this purpose. One common method involves using a primary antibody that is conjugated to an enzyme, such as horseradish peroxidase (HRP) or alkaline phosphatase (AP). These enzymes catalyze a reaction that produces a visible colorimetric signal, allowing the localization of the antigen of interest. Alternatively, immunofluorescence (IF) staining can be performed, in which a primary antibody is tagged with a fluorophore such as fluorescein, rhodamine, or Alexa Fluor, and the fluorescence is detected using a specialized microscope.
Another approach involves using an unlabeled primary antibody, followed by detection with a labeled secondary antibody. The secondary antibody binds to the primary antibody, and the detection of the secondary antibody is typically achieved through the use of fluorescent labels, enzyme conjugates, or other complex detection systems. In such cases, it is important to determine the optimal titer for both the primary and secondary antibodies to ensure the specificity and sensitivity of the assay.