22-181-000
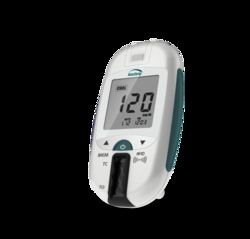
LumiraDx Instrument for Rapid Detection of SARS-CoV-2
Manufacturer: LumiraDx
Select a Size
| Pack Size | SKU | Availability | Price |
|---|---|---|---|
| Each of 1 | 22-181-000-Each-of-1 | In Stock | ₹ 3,94,537.00 |
22-181-000 - Each of 1
In Stock
Quantity
1
Base Price: ₹ 3,94,537.00
GST (18%): ₹ 71,016.66
Total Price: ₹ 4,65,553.66
Sample Type
Proteins from SARS to CoV to 2
Description
Used with LumiraDx SARS-CoV-2 Ag Test
Format
Portable instrument
Description
- Features: Innovative, next generation point of care system that combines a small, portable instrument, advanced low-cost test strip and seamless digital connectivity Delivers exceptional point of care clinical performance Simple and easy to use Performance referenced to laboratory test Portable instrument, seamless connectivity One simple, intuitive test workflow Fast results in minutes Nasal swab specimen type Room temperature storage Used with the LumiraDx SARS-CoV-2 Ag Test Notes: Orders require executed contractual agreement Please contact your Fisher Healthcare Account Representative for more information This test has not been FDA cleared or approved; but has been authorized by FDA under EUA for use by authorized laboratories; use by laboratories certified under the CLIA, 42 U.S.C
- §263a, that meet requirements to perform moderate, high or waived complexity tests Test authorized for use at the Point of Care (POC), i.e., in patient care settings operating under a CLIA Certificate of Waiver, Certificate of Compliance, or Certificate of Accreditation Authorized only for the detection of proteins from SARS-CoV-2, not for any other viruses or pathogens In USA, test is only authorized for duration of declaration that circumstances exist justifying the authorization of emergency use of in vitro diagnostics for detection and/or diagnosis of the virus that causes COVID-19 under Section 564(b)(1) of the Federal Food, Drug, and Cosmetic Act, 21 U.S.C
- § 360bbb-3(b)(1), unless authorization is terminated or revoked sooner