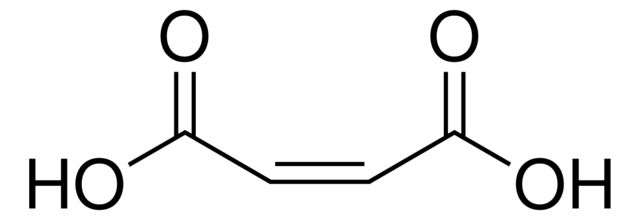
Maleic acid
electronic grade, ≥99.97% trace metals basis, water <=350 ppm
Manufacturer: Sigma Aldrich
CAS Number: 110-16-7
Synonym(S): (2Z)-2-Butenedioic acid, (2Z)-But-2-enedioic acid, Maleinic acid, cis-Butenedioic acid, Toxilic acid
Select a Size
| Pack Size | SKU | Availability | Price |
|---|---|---|---|
| 500 G | 912735-500-G | In Stock | ₹ 5,845.50 |
912735 - 500 G
In Stock
Quantity
1
Base Price: ₹ 5,845.50
GST (18%): ₹ 1,052.19
Total Price: ₹ 6,897.69
grade
electronic grade
Quality Level
100
Assay
≥99.97% trace metals basis
form
powder
color
white to off-white
mp
130-135 °C (lit.)
density
1.59 g/mL at 25 °C (lit.)
SMILES string
OC(=O)\C=C/C(O)=O
InChI
1S/C4H4O4/c5-3(6)1-2-4(7)8/h1-2H,(H,5,6)(H,7,8)/b2-1-
InChI key
VZCYOOQTPOCHFL-UPHRSURJSA-N
Other Options
| Image | Product Name | Manufacturer | Price Range | |
|---|---|---|---|---|
Quality Environmental Containers Maleic/Ascorb Acid | 110-16-7 | MFCD00063177 | 40 mL | -- | ₹ 20,462.88 | ||
Medchemexpress LLC HY-Y0367 500mg Medchemexpress, Maleic Acid CAS:110-16-7 Purity:>98% | -- | ₹ 3,284.10 | ||
Maleic acid Pharmaceutical Secondary Standard, MilliporeSigma™ Supelco™ | -- | ₹ 11,623.40 | ||
U.S. Pharmacopeia Maleic acid, 110-16-7, MFCD00063177, 300 mg | -- | ₹ 90,632.26 | ||
Sigma Aldrich Fine Chemicals Biosciences Maleic Acid | 110-16-7 | MFCD00063177 | 1KG | -- | ₹ 9,267.57 | ||
Sigma Aldrich Fine Chemicals Biosciences Maleic Acid | 110-16-7 | MFCD00063177 | 100g | -- | ₹ 7,805.30 | ||
Maleic acid | -- | ₹ 17,655.58 | ||
Maleic acid | -- | ₹ 9,645.08 | ||
Maleic acid | -- | ₹ 1,29,412.88 | ||
Maleic acid | -- | ₹ 16,107.60 |
Description
- General description: Maleic acid is the cis-isomer of butenedioic acid, an unsaturated organic dibasic acid. Maleic acid shows reactions typical of both olefins and carboxylic acids. Commercially important reactions of the acid groups include esterification with glycols to polyesters and dehydration to the anhydride. The double bond is involved in conversions to fumaric acid, to sulfosuccinic acid, and to Malathion.
- Application: Maleic acid has been adopted as a high performance organic anode material for lithium ion batteries. In one study, it showed an ultrahigh reversible capacity of ca. 1500 mAhg–1 at 46.2 mAg–1 current density, and a relatively high capacity of 570.8 mAhg–1 at current density of 46.2 Ag–1. When cycled at high current density of 2.31 Ag–1, a capacity retention of 98.1% was obtained after 500 cycles.[1]Maleic acid has been used in constructing polymeric solid electrolytes for battery and fuel cell. In one study, it was showen that maleic acid based polymer blend helps increasing the ionic conductivity of PEO basd polymer solid electrolytes by suppressing its crystallization.[2]Maleic acid has also been used to make superswelling acrylamide (AAm)/maleic acid (MA) hydrogels by free radical polymerization in aqueous solution of AAm with MA as comonomer with some multifunctional crosslinkers such as trimethylolpropane triacrylate and 1,4-butanediol dimethacrylate. When the uptake of cationic dye was studied, it was shown that the binding ratio of hydrogel/dye systems was gradually increased with the increase of MA content in the AAm/MA hydrogels.[3]
SAFETY INFORMATION
Pictograms
Signal Word
Danger
Hazard Statements
Precautionary Statements
P260 - P271 - P280 - P301 + P312 - P303 + P361 + P353 - P305 + P351 + P338
Hazard Classifications
Acute Tox. 4 Dermal - Acute Tox. 4 Oral - Eye Dam. 1 - Skin Corr. 1 - Skin Sens. 1 - STOT SE 3
Target Organs
Respiratory system
WGK
WGK 1