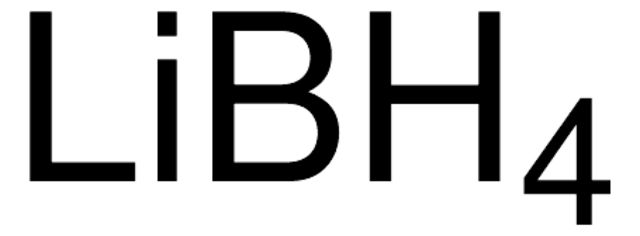452882
Sodium borohydride
powder, ≥98.0%
Manufacturer: Sigma Aldrich
CAS Number: 16940-66-2
Synonym(S): Sodium tetrahydridoborate
Select a Size
| Pack Size | SKU | Availability | Price |
|---|---|---|---|
| 5 G | 452882-5-G | In Stock | ₹ 7,220.28 |
| 25 G | 452882-25-G | In Stock | ₹ 7,609.98 |
| 100 G | 452882-100-G | In Stock | ₹ 10,023.95 |
| 6 X 25 G | 452882-6-X-25-G | In Stock | ₹ 10,424.48 |
| 6 X 100 G | 452882-6-X-100-G | In Stock | ₹ 20,058.73 |
| 500 G | 452882-500-G | In Stock | ₹ 24,074.80 |
| 2 KG | 452882-2-KG | In Stock | ₹ 84,337.58 |
452882 - 5 G
In Stock
Quantity
1
Base Price: ₹ 7,220.28
GST (18%): ₹ 1,299.65
Total Price: ₹ 8,519.93
Quality Level
300
Assay
≥98.0%
form
powder
contains
0.45-0.70% Magnesium carbonate as anticaking agent
reaction suitability
reagent type: reductant
greener alternative product characteristics
Design for Energy EfficiencyLearn more about the Principles of Green Chemistry.
sustainability
Greener Alternative Product
impurities
≤100 ppm Silica
mp
>300 °C (dec.) (lit.)
cation traces
Fe: ≤5 ppm
Other Options
Description
- General description: Sodium borohydride (NaBH4) is the most commonly available borohydride, synthesized by reacting methylborate with sodium hydride in mineral oil. It is employed as a precursor for producing other metal borohydrides. Various approaches for qualitative and quantitative monitoring of sodium borohydride have been explained.[1] The potential of NaBH4 to store as well as generate hydrogen for fuel cells has been investigated.[2][3]
- Application: Sodium borohydride can be used as a reducing agent: To prepare saturated alcohols by the reduction of conjugated aldehydes and ketones.[4] Along with Raney nickel for the reduction of aromatic nitro compounds to arylamines.[5] In the synthesis of secondary amines by reductive amination of ketones and aldehydes in the presence of an acid catalyst.[6] In the synthesis of δ and γ lactones by the reduction of cyclic anhydrides.[7] For the chemoselective reduction of N -protected amino acids and peptides to corresponding alcohols.[8]
- Legal Information: ASCENSUS SPECIALTIES LLC
SAFETY INFORMATION
Pictograms
Signal Word
Danger
Hazard Statements
Precautionary Statements
P231 + P232 - P260 - P280 - P303 + P361 + P353 - P304 + P340 + P310 - P305 + P351 + P338
Hazard Classifications
Acute Tox. 3 Oral - Eye Dam. 1 - Repr. 1B - Skin Corr. 1B - Water-react 1
Supplementary Hazards
EUH014
WGK
WGK 2
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves