1.00894
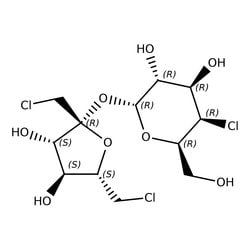
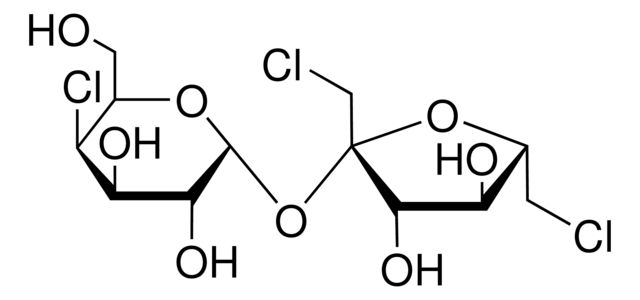
Sucralose
powder, EMPROVE® ESSENTIAL, Ph. Eur., ChP, JPE, NF
Manufacturer: SAFC
CAS Number: 56038-13-2
Synonym(S): Sucralose, 1,6-Dichloro-1,6-dideoxy-β-D-fructofuranosyl-4-chloro-4-deoxy-α-D-galactopyranoside, E955, Trichlorosucrose
Select a Size
| Pack Size | SKU | Availability | Price |
|---|---|---|---|
| 10 G | 1.00894-10-G | In Stock | ₹ 4,886.41 |
| 1 KG | 1.00894-1-KG | In Stock | ₹ 71,978.68 |
| 10 KG | 1.00894-10-KG | In Stock | ₹ 7,34,573.68 |
1.00894 - 10 G
In Stock
Quantity
1
Base Price: ₹ 4,886.41
GST (18%): ₹ 879.554
Total Price: ₹ 5,765.964
Agency
ChPJPENFPh. Eur.
Quality Level
500
description
EMPROVE® ESSENTIAL Ph Eur,NF,JPE
product line
EMPROVE® ESSENTIAL
form
fine powderpowder
particle size
≤12 μm (d90)
pH
6-8 (20 °C, 100 g/L in H2O)
mp
130 °C (decomposition)
density
1.62 g/cm3 at 20 °C
application(s)
liquid formulationpharmaceuticalsolid formulation
Other Options
| Image | Product Name | Manufacturer | Price Range | |
|---|---|---|---|---|
 | eMolecules Ambeed / (2R3R4R5R6R)-2-(((2R3S4S5S)-25-Bis(chloromethyl)-34-dihydroxytetrahydrofuran-2-yl)oxy)-5-chloro-6-(hydroxymethyl)tetrahydro-2H-pyran-34-diol / 5g / 506391275 / A551773 / / 56038-13-2 / MFCD03648615 / 397.630 / C12H19Cl3O8 | eMolecules | ₹ 1,978.15 | |
 | Sucralose Pharmaceutical Secondary Standard, MilliporeSigma™ Supelco™ | MilliporeSigma Supelco | ₹ 11,430.82 | |
 | Sigma Aldrich Fine Chemicals Biosciences Sucralose European Pharmacopoeia (EP) Reference Standard | 56038-13-2 | MFCD03648615 | | Sigma Aldrich Fine Chemicals Biosciences | ₹ 23,582.90 | |
 | Sucralose | Supelco | ₹ 10,695.10 | |
 | Sucralose | Supelco | ₹ 16,421.53 | |
 | Sucralose | Sigma Aldrich | ₹ 51,916.70 | |
 | Sucralose | Sigma Aldrich | ₹ 20,567.50 | |
 | Sucralose | Sigma Aldrich | ₹ 19,008.70 | |
 | Sucralose | SAFC | ₹ 4,886.41 - ₹ 6,14,167.20 | |
 | Sucralose | Sigma Aldrich | ₹ 19,008.70 |
Description
- General description: Finding the right excipient that matches your needs as well as regulatory demands can be a complicated challenge in formulation. With our application know-how and regulatory expertise, we support you in every step of development, scale-up, and production.As part of our EMPROVE® Program, our raw materials are offered with extensive documentation facilitating compliance of your pharma and biopharma product, full supply chain transparency and risk mitigation. Our SAFC® portfolio of high-quality products for biopharmaceutical and pharmaceutical formulation and production withstands strict quality control procedures and is produced according to applicable cGMP guidelines.
- Application: Sucralose is a high-intensity sweetener which is non-cariogenic and suitable for diabetics and patients with an impaired glucose tolerance. In addition, it is readily soluble and masks both bitterness and medicinal off-notes with equal ease – making it exceptionally well-suited for pharmaceutical applications.Our Sucralose is available in micronized or granular form, both pharmaceutical grade, allowing you to choose depending on your individual needs. Due to its optimal dispersion behavior, sucralose powder is typically recommended for solid dose formulations, while the fast dissolution behavior of sucralose granular makes it very well-suited for liquid formulations.
- Biochem/physiol Actions: A synthetic sweet tastant detectable by humans. Activates T1R2/T1R3 sweet taste receptors on enteroendocrine cells and elicits increased hormonal secretion of glucagon-like peptide-1 and glucose-dependent insulinotrophic peptide.
- Features and Benefits: Excellent sugar-like taste profiles Masking bitter aftertaste and medical off-notes with ease High sweetness potency offers improved cost-efficiency Enhanced stability Non-cariogenic and suitable for diabetics and patients with phenylketonuria
- Legal Information: Emprove is a registered trademark of Merck KGaA, Darmstadt, Germany
- Disclaimer: The substance 1.00894 has no REACH registration because the only supported use is the use in medicinal products for human or veterinary use or in food or feedingstuffs according to article 2 of the REACH Regulation (EC) No 1907/2006.
SAFETY INFORMATION
WGK
WGK 2
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Compare Similar Items
Show Difference
Agency: ChPJPENFPh. Eur.
Quality Level: 500
description: EMPROVE® ESSENTIAL Ph Eur,NF,JPE
product line: EMPROVE® ESSENTIAL
form: fine powderpowder
particle size: ≤12 μm (d90)
pH: 6-8 (20 °C, 100 g/L in H2O)
mp: 130 °C (decomposition)
density: 1.62 g/cm3 at 20 °C
application(s): liquid formulationpharmaceuticalsolid formulation
Agency:
ChPJPENFPh. Eur.
Quality Level:
500
description:
EMPROVE® ESSENTIAL Ph Eur,NF,JPE
product line:
EMPROVE® ESSENTIAL
form:
fine powderpowder
particle size:
≤12 μm (d90)
pH:
6-8 (20 °C, 100 g/L in H2O)
mp:
130 °C (decomposition)
density:
1.62 g/cm3 at 20 °C
application(s):
liquid formulationpharmaceuticalsolid formulation